Originally posted on Jabil Blog
By Bob Barnhart, Director, Business Development, Jabil Healthcare
Transferring manufacturing work is like a trust fall. It’s one of the biggest tests in the relationship between an original equipment manufacturer (OEM) and a manufacturing solutions provider. As they close their eyes and lean back, OEMs place their trust in their new or established partners. Will the manufacturing solutions provider or contract manufacturer be there to catch them?
 As the healthcare industry’s largest manufacturing solutions provider, Jabil has extensive experience transferring manufacturing operations from customers, and even competitors, to our facilities. In fact, we leverage a multi-disciplinary, collaborative approach to helping customers improve product safety, performance, sustainability and accessibility.
As the healthcare industry’s largest manufacturing solutions provider, Jabil has extensive experience transferring manufacturing operations from customers, and even competitors, to our facilities. In fact, we leverage a multi-disciplinary, collaborative approach to helping customers improve product safety, performance, sustainability and accessibility.
For instance, in our work supporting Candela, a leader in medical aesthetic devices, one of the first steps we took was to consolidate Candela’s manufacturing and supply chain operations.
Jabil’s Director of Business Development, John Vennari, worked hand-in-hand with Candela to understand their products and prepare them for transfer. “Candela had multiple programs moving from multiple locations. We had to take a very careful view of the planning, supply chain and testing issues to ensure a smooth transfer,” he said.
Faced with extremely aggressive timelines, Jabil quickly mobilized highly qualified transfer teams to move Candela’s current manufacturing and complex capital equipment to a Jabil Healthcare facility in Tijuana, Mexico. Download the Candela Case Study.
“Through the transfer process, Jabil was able to catalyze its expertise,” Candela’s Chief Operating Officer Todd Van Horn said. “They guided innovation in terms of process improvements, manufacturing best practices and quality.”
For myself, with 15 years at Jabil and 20 years in the military and defense sector, I have repeatedly seen the benefits of careful planning for a manufacturing transfer. Conversely, I’m aware of the trouble caused by delays, when preparation is not as careful as it needs to be.
As they say, the devil is in the details with a manufacturing transfer—especially with medical products where the bar is set so high due to strict regulatory requirements. With the healthcare sector going through such a dynamic phase, it’s a good time to walk through four considerations for those planning a manufacturing transfer to ensure a safe pair of hands catches your “fall:” planning, communications, materials and testing.
Manufacturing Transfer Checklist: Four Considerations to Get it Right
1. Planning is Vital to a Manufacturing Transfer
Developing a robust, well-defined project plan document is listed first since it’s arguably the most important step in the manufacturing transfer. If you can create a comprehensive project plan and schedule with clear ownership, you are more than halfway to a successful transfer. The plan should be developed by experienced Project Managers (PMs) – on both the customer and manufacturer side – with clear roles and accountability defined.
Your project management team is vital to a successful transfer of your manufacturing process. The PMs are the conductors of this orchestra and must be empowered to ensure that everything is in order before they lift the baton for the first note. Everyone on the team should know their responsibilities. Each side of the partnership relies on the other to understand all the elements required for overall program success.

2. Consistent Communication is Key
Communication is the next critical leg for a successful manufacturing transfer. While planning gets you up and running, the baton is passed through a comprehensive communication strategy – one that is both consistent and transparent. As we’re seeing today, disruptions of medical equipment inventory to hospitals and other care facilities don’t just impact patients’ quality of life, it can mean the difference between life or death.
Healthcare products are often composed of sensitive componentry with intricate design geometries. Combining these manufacturing challenges with the industry’s complex regulatory requirements necessitates that all the details of a product’s supply chain, history and testing be well-known to the manufacturer.
The most successful manufacturing transfer programs have strong, empowered PMs who work together as partners – ensuring that all aspects of the program are documented, scheduled and understood. As the project kicks off, this communication must continue to be open and comprehensive – working together regularly for the best outcome. The following table outlines recommendations for good communication throughout the program — from initial planning through regular production.
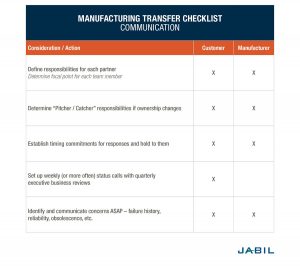
3. Track All Materials During a Manufacturing Transfer
Roadmaps are an essential navigational tool for ensuring manufacturing transfer success by recording where materials are coming from and where they are going. Even as manufacturing old-timers muse “we just make stuff,” products are more sophisticated than ever. It takes “stuff” to make “stuff,” and being able to locate, track and account for the flow of it all is critical to keeping production on time and on budget.
Every part and piece that goes into a product must be accounted for prior to kick off. Take a detailed inventory of all required materials, including their costs, their lifecycle stages and their supply chains. And be sure to also include your suppliers’ inventories and capabilities, as well.
As technology works its way into more and more products, the consideration of end-of-life (EOL) becomes increasingly critical. Frequent iterations and upgrades in technology are the norm, which impacts your supply chain downstream. The discovery and hassle of an EOL part is happening more frequently as product lifecycles speed up across the board.
Here are four vital areas of consideration for materials transfer – inventory, supplier pricing, supplier lifecycle status and viability. The following checklist outlines important manufacturing transfer considerations relating to materials.
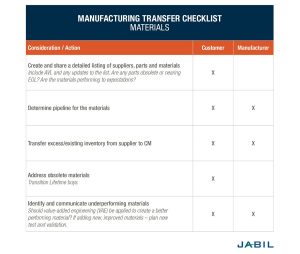
4. Establish Testing & Validation Ownership
“Who owns the testing?”
When undertaking a transfer project from the OEM to a manufacturing partner, this must be one of the first questions asked and answered. Testing processes and procedures must meet validation and compliance standards. These concerns are certainly foundational in all industries, but in healthcare, as noted before, they could mean the difference between life and death.
In most cases, ownership of the test solution belongs to the customer, but there are circumstances where it falls under the control of a partner. It doesn’t matter which; the point is that just getting to the answer is a requirement. Nothing should be left to chance. If there is a change in materials or suppliers, those pieces and parts have to be validated, so it’s crucial that the project managers build in enough time to account for re-testing.
The following table outlines important considerations and possible scenarios to ensure testing and process validation are up to regulatory standards for a finished product during a manufacturing transfer.

Built upon the cornerstone of a company-wide dedication to operational excellence, Jabil is proud of our outstanding track record executing new product introductions and technology transfer programs. Decades of experience have reinforced the importance of detailed project planning, dependency analysis and transparent communications.
Getting it right upfront is the only way to get it right overall. With extensive collaboration and built-in trust, these types of partnerships can help you mitigate risk and better prepare for the unexpected with flexibility. Are you ready for this trust fall?













